Introduction
The global egg industry produces more than a million tons of eggs each year, making a significant contribution to the world’s protein supply and supporting global nutrition and food security. Egg white (EW) proteins are commonly used in various processed foods (Herawati et al., 2021; Zhang et al., 2013). Enhancing the natural gelling properties of egg proteins could lead to an improved quality of these processed foods (Li et al., 2021a). Freezing egg products for preservation is a popular practice, but it can lead to texture deterioration due to syneresis, which depends on storage conditions and ingredients (Chevalier et al., 2000; Eregama et al., 2024; Sen et al., 2021).
The leaves of olive trees (Olea europaea L., Oleaceae) contain high levels of phenolic compounds, with the primary phenolic compound being oleuropein at 10.9–12.6 g/100 g dry weight of olive leaf (Ohyama et al., 2016). Oleuropein is specific to plants in the Oleaceae family and is not found in plants from other families. In addition to oleuropein, olive leaves also contain other major phenolics such as hydroxytyrosol and verbascoside, which are structurally different from the flavonoids found in the leaves of plants from other families, such as theaceae and vitaceae. Olive leaves also contain oxidative enzymes, including peroxidase and polyphenol oxidase, which play a role in converting phenolic compounds into quinone forms (De Leonardis et al., 2015; Motamed et al., 2007; Ortega-García et al., 2008).
The enhancement of the gel properties can be attributed to the promotion of intermolecular cross-linking of proteins by phenolic compounds. This process leads to a more stable and structured gel network, resulting in improved performance and functionality. Phenolic compounds can interact with proteins through both non-covalent and covalent interactions (Prigent et al., 2003). Covalent interactions, in particular, have been extensively studied for their role in modifying the physical and chemical properties of protein gels. When phenolic compounds with an ortho-diphenol structure are oxidized, they form a covalent bond with proteins, leading to the formation of cross-linked protein polymers (Strauss and Gibson, 2004). This process enhances the strength of protein gels by creating crosslinks among protein chains.
Recent studies have shown that adding plant extracts or phenolic compounds to food protein hydrogels can alter the physical properties of egg gels. For instance, the addition of tea extract containing catechins has been found to enhance the breaking strength of EW gel (Hatanaka et al., 2009; Wu et al., 2007). Akazawa et al. (2021) have suggested that using olive leaf extract, prepared with cold water from olive leaves without blanching treatment, can improve the viscoelasticity, breaking stress, and water-holding capacity (WHC) of EW gel. This additive helps to maintain the texture of egg gel products during frozen storage. Additionally, according to Rachman et al. (2021), olive leaf extract can significantly reduce the deterioration of the rheological properties of sausages during frozen storage, thereby improving the preservation stability of meat products in frozen foods.
As the frozen food market expands, many gel-like processed foods must be equipped with high frozen resistance, particularly the anti-syneresis effect. This is because defrosting frozen foods causes syneresis, resulting in the deterioration of texture. There is no data on the anti-syneresis effect or frozen resistance of egg gels treated by chemical modifications such as glycation and the addition of polyphenols. In our research, we examined the properties of egg gels following freeze-thaw cycles to evaluate the freeze resistance of egg gels that have been fortified with an extract from olive leaves.
Materials and Methods
The materials used in the study included fresh olive leaves (O. europaea L.) sourced from the Agriculture Faculty of Kagawa University in Japan. The eggs (Boris Brown hen strain) were purchased from a retail market in Hyogo Prefecture, Japan. Kureha (Tokyo, Japan) provided the Krehalon® PVDC casing used as a handmade egg gel stuffer. Analytical-grade chemicals were used for all compounds.
Olive leaves were dried for 48 h at a temperature between 25°C and 40°C in a cold-air drying machine (Cool Dry Machinery, Kagawa, Japan) and subsequently pulverized. The ensuing powder was filtered through a nylon mesh sieve with a pore diameter of 108 μm. Two extracts of olive leaf water were prepared from the sieved olive leaf powder at a temperature of 4°C. An Erlenmeyer flask containing 500 mL of pre-cooled, 4°C distilled water and 50 g of olive leaf powder was swirled for an hour at 4°C using a magnetic stir plate. Centrifugation 8,000×g and 4°C was performed for 20 min on the homogenates obtained from this procedure. Lyophilization was applied to the supernatant and olive leaf water extract (OEx4) was made from the resulting lyophilized powder. OEx4 was kept in storage at –20°C, until it was needed for additional testing.
The EW gel and whole egg (WE) gels were prepared from fifty hen eggs each. EW was divided from egg yolk (EY) using an egg separator, and the EW was gently collected in a container. WE, after breaking eggs, was gently collected in a separate container. Each EW and WE solution was mixed with a Nissei AM-8 homogenizer (Nihonseiki Kaisha, Tokyo, Japan) was used to stir. Subsequently, either 0.03 or 0.1 g of OEx4 was combined with 100 mL of the EW or WE solution. The egg solution was put into a Krehalon® PVDC casing (Kureha, Tokyo, Japan), which had a flat width of 4.0 cm. The casing was heated for 30 min at 80°C. Post-heating, the resulting cylindrical gel was approved to rest at room temperature for one hour and was then refrigerated at 4°C for 6 h. Each cylindrical egg gel (with a diameter of 17 mm) was then removed from its casing and sectioned at 20 mm intervals using a razor blade for the purpose of the freeze-thaw experiment.
Cylindrical egg gels were utilized to assess how OEx4 affected the freezing and thawing process. Twenty-one sets of cylindrical gels were stored in a lidded polypropylene plastic box, which was kept for five days at –20°C in a still-air freezer (Biomedical Freezer, MDF-436; Sanyo, Osaka, Japan). The frozen gels were then defrosted in a refrigerator (4°C) for 16 h. After being freeze-thawed, seven cylindrical gel samples were physically inspected. Two or three more freeze-thawing steps were done, and physical analyses were conducted on the remaining 14 gel samples.
An aluminum foil pan was filled with 2 g of egg gel pieces. Following that, these pieces were dried in a drying cabinet set at 105±1°C until the sample’s weight remained constant. The dehydrated sample was chilled in a desiccator containing silica gel prior to being measured using an precision balance. The water content in the egg gel can be determined using the appropriate formula:
where, Wf denotes the egg gel’s final weight following dehydration and Wi denotes the egg gel’s weight before dehydration.
The WHC method was used to quantify the amount of expressible water in the egg gel with slight modifications (Visessanguan et al., 2004). Utilizing a Rheoner II creep meter (model RE 2-3305; built by Yamaden, Tokyo, Japan) fitted with a 40 mm diameter cylindrical probe, an egg gel cylinder measuring 17.5 mm in diameter and 10 mm in height was squeezed. The compression was carried out at a crosshead speed of 5 mm/s to 50% strain for a duration of 60 s. The expressible water of the egg gel was determined by applying the subsequent formula:
Here, Wi represents the slice of egg gel’s weight at initial contact, and Wc represents the weight of the condensed area.
The egg gel’s thawing loss was measured using the method of thawing loss with minor modifications (DeFreitas et al., 1997). The following formula was utilized to calculate the thawing loss of the frozen cylindrical gel samples:
In this equation, Wi is the weight of a cylindrical gel before it is frozen, and Wf is the weight of the gel after it has been thawed.
A pH meter was used to determine the egg gel’s pH level (model EutechTM PC 700 Multi-parameter Meter, manufactured by Thermo Fisher Scientific, Waltham, MA, USA). This measurement was taken after 3.0 g of the egg gel was homogenized with 27 mL of distilled water using a Digital High Shear D-160 (produced by BIOBASE, Wolfenbuettel, Germany) at a speed of 3,396×g for a duration of 30 s at room temperature.
Microbiological study of the samples was done in cycles (0, 1, 2, and 3) of frozen storage, with seven replicates per group. That is, at each time point, 21 samples (2 concentrations of olive leaf extract+control) were examined. The total plate count (TPC) was determined with Plate Count Agar (PCA, Merck, Darmstadt, Germany) incubation at 37.8°C for 24 h and enumerated. All counts of TPC from egg gel sample are expressed as Log CFU/g.
The viscoelastic property and the egg gel’s breaking strength was evaluated using a Rheoner II creep meter. A creep-recovery test was conducted at 25°C to assess the viscoelastic property. The cylindrical gel, which had a diameter of 17.5 mm, was subjected to a continuous tension of 120 N/m2 and a height of 10 mm for 1 min at a crosshead speed of 5 mm/s using a plate plunger with a diameter of 40 mm, in order to estimate creep compliance. Following this, the stress was released for 1 min to obtain the stress compliance curve of recovery. The creep-recovery curve was assessed with the use of six-element mechanical model and software provided by the manufacturer (CAS-3305, ver. 1.6). The instantaneous modulus (E0) of a Hookean body and the Newtonian viscosity (ηN), two key quantities, were identified. Using a cylindrical plunger with a 3 mm diameter. 1.0 mm/s of penetration speed was used to create a stress-strain curve. The stress (N/m2) and strain (%) at the peak of the initial stress-strain curve were used to represent the breaking stress and strain.
Differential scanning calorimetry (DSC) was used to identify the degree of heat denaturation of the proteins in the egg batter. For the DSC analysis, a Setaram Micro-DSC VII Commissioning/Utilizations instrument (produced by SETARAM Instrumentation, Caluire, France) was used. Two hundred milligrams of Al2O3 and egg batter were added to a reference pan and a sample pan, respectively. The samples were heated from 20°C to 100°C at a rate of 1.2°C per second. The thermal analysis software included with the DSC device was used to examine the calorimetric data.
The microstructure of the egg gel was examined using scanning electron microscopy. After being sliced into 2–3 mm pieces thick using a knife, the egg gel pieces were fixed for 2 hours at 4°C with 2.5% glutaraldehyde in a 100 mM sodium phosphate buffer (pH 7.4). They were then post-fixed with 2% osmium in the same solution. Following fixation, the sample underwent 10 min of ethanol dehydration at increasing concentrations [50%, 70%, 80%, 90%, 95%, and 100% (v/v)], followed by freeze-drying. After being dehydrated, the sample was attached to a bronze stub and covered in gold (DII-29010SCTR, JEOL, Tokyo, Japan). Subsequently, it was examined using a scanning electron microscope (JCM-6000, JEOL) operating at an acceleration voltage of 15 kV.
OEx4, in quantities of 0.3 and 1.0 mg, was dissolved in 1.0 mL of a 10 mM sodium phosphate buffer (pH 7.0) and subsequently combined with 1.0 mL of a 10 mg/mL egg (WE or EW) protein solution. The egg protein solution, once mixed with OEx4, was subjected to heat treatment at 80°C for a duration of 30 min. The dissociation solution (8 M urea and 12.5% SDS) was added in a volume four times larger than the hazy solution that was produced. The protein was dissolved in the dissociation solution by vortexing for 4 min, and the sample was then processed for sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE). The specimen was combined with a 4×SDS-PAGE sample buffer, which consisted of a pH 6.7 100 mM Tris-HCl buffer, 2% β-mercaptoethanol, 0.004% bromophenol blue, and 5% SDS) then heated for two min at 100°C. An SDS-PAGE gel (12.5% acrylamide separating gels) was loaded with a 10 μL aliquot of the prepared sample solution. Following electrophoresis, the gel underwent staining with a 0.025% solution of Coomassie Brilliant Blue R250.
The experiment results were obtained from a completely randomized design of at least three independent replicates for each treatment. Statistical analysis was carried out using SPSS 23.0 software by performing a one-way analysis of variance (ANOVA) and the two-way ANOVA in some experiments. The statistical difference of the mean was ascertained using the Duncan multiple range test at a 95% significant level. The data are reported as mean and SD.
Results and Discussion
This study investigated how the addition of OEx4 affects the gel properties of egg gels during the freeze-thaw process. The study compared the gel properties of WE gel with 0.03% and 0.1% OEx4 (Table 1). The results showed that adding OEx4 to the WE gel resulted in a water content similar to the control, with no significant difference (p>0.05). However, the expressible water of the WE gel decreased significantly with the addition of OEx4 (p<0.05), indicating that OEx4 improves the gel’s ability to hold water. The breaking stress, elongation, elasticity, and viscosity of WE gels increased with the addition of OEx4 (p<0.05). Thus, OEx4 enhances the gel properties of WE gel. Based on the Table 2, the gel properties of EW gel containing OEx4 and found that the water content did not change (p>0.05). However, the expressible water decreased significantly with the addition of OEx4 (p<0.05), indicating an improvement in the gel’s WHC. Importantly, the study found that adding OEx4 to WE or EW solution at 0.03% (w/v) is sufficient, as the impact did not significantly differ from that of 0.1% OEx4.
Adding OEx4 improves the elasticity and strength of WE and EW gels by altering the physical properties of the eggs with natural enzymes. The interaction between polyphenol and protein molecules leads to irreversible effects, typically occurring through C-N or C-S linkage (Li et al., 2021b). This process commences with the formation of quinones, which can arise through enzymatic modification in the presence of oxygen or through autoxidation under alkaline conditions (Shahidi and Dissanayaka, 2023). Ozdal et al. (2013) explained that polyphenol oxides help in converting monophenols into ortho-diphenols through hydroxylation, and then rapidly oxidizing them into ortho-quinones. These quinones, which are strong electrophiles, react with nucleophilic residues of proteins or peptides (such as thiol, amino, guanidine, or imidazole) through Schiff base (C=N) and Michael addition mechanisms (C–NH), potentially leading to the formation of protein cross-links. Balange and Benjakul (2009) demonstrated that oxidized phenolic compounds such as ferulic acid, tannic acid, catechin, and caffeic acid improve the WHC of surimi gel, and further research has shown that the oxidation of these polyphenols results in the cross-linking of myofibrillar proteins in surimi. Handa et al. (2001) also suggested a strong relationship between WHC and the physical properties of heat-induced gels of EWs.
Two stages thermally induced gelation were used for the egg proteins. The protein is first heated to a denaturation point. The rate of protein diffusion increases when proteins become denatured (Alleoni, 2006). The second stage involves interactions between denatured protein molecules and other denatured molecules. Proteins that interact for extended periods of time form high molecular weight aggregates. As the aggregates continue to form, they create an elastic gel that can hold water molecules. Thus, understanding the gel characteristics of EY requires a thorough examination of protein denaturation and protein-protein interactions. A single endothermic peak was observed within the temperature range of 82.09°C to 82.34°C, according to Fig. 1A for all WE samples. In the meantime, all EW samples showed two sizable endothermic peaks (Fig. 1B). The first peak shows in the range of 64.15°C and 64.41°C, and the second peak at 82.15°C and 82.48°C (Renzetti et al., 2020). Two endothermic peaks may be seen in the denaturation of ovalbumin and ovotransferrin in the EW (Gorinstein et al., 1995). The two highest points are 63.6°C and 76.18°C (Sun et al., 2006). Thus, ovotransferrin was associated with the first peak in the EW thermogram, and ovalbumin was associated with the second and sole peak in the WE thermogram. The peak in WE and EW thermograms changed to higher temperatures at 0.25°C or 0.33°C when olive leaf extract was added. This implies that the heat-induced denaturation of egg proteins is slowed down by olive leaf extract. EY development depends on ovalbumin, the most common protein in eggs (Razi et al., 2023). When olive leaf extract was present, the denaturation temperature of ovalbumin was relatively close to that of ovalbumin without extract. The characteristics of the gels with and without olive leaf extract changed noticeably as a result of the temperature difference. The appropriate gel binding of EYs with olive leaf extract depends on the larger temperature shift of ovalbumin denaturation temperature caused by the extract. To ensure that EYs containing olive leaf extract are properly bound, the ovalbumin denaturation temperature must be raised to a higher degree.
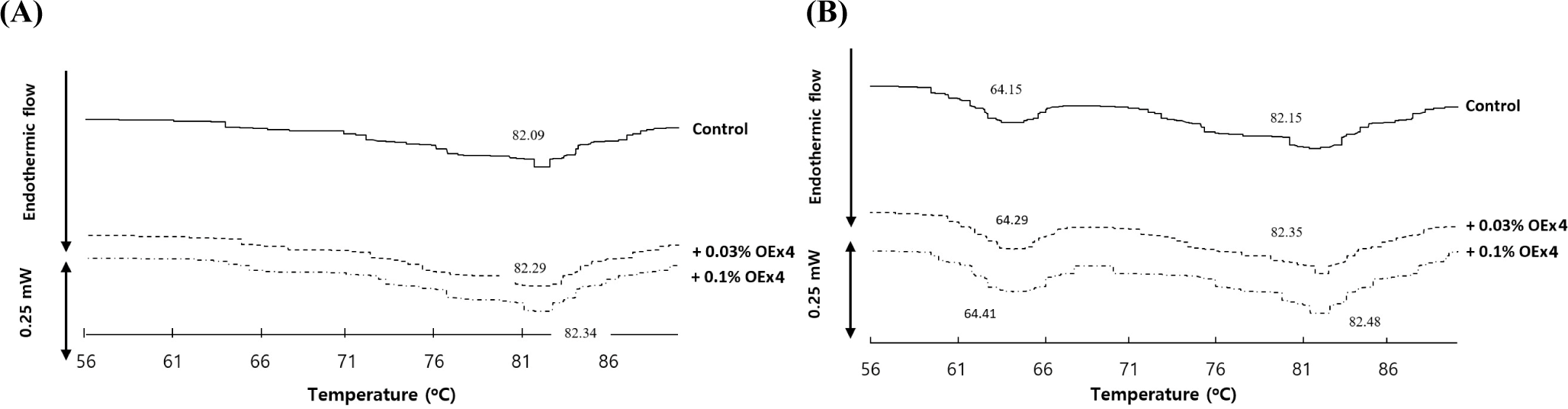
Fig. 2 illustrates that both WE and EW gels experienced drip and thawing losses when frozen, leading to a decrease in food viscosity and taste. The thawing loss of the control gel sample is significantly increased by repeating the freeze-thaw cycle (p<0.05), while that of OEx4-gel pieces was stable (p>0.05). The freeze-thaw result of WE and EW gels suggests that OEx4 addition effectively prevents the syneresis from proteinous food gels by frozen storage. The oxidized polyphenols present in OEx4 create robust cross-links with the proteins in egg gel. This interaction plays a crucial role in inhibiting the formation of ice crystals during the freezing process. As a result, the drip loss, which refers to the loss of moisture from the gel, is not reduced after freezing. Polyphenols are natural antioxidants that provide excellent protection against oxidation by neutralizing free radicals (Huang et al., 2019). Elgamouz et al. (2019) discovered that natural antioxidants such as α-Tocopherol, cinnamon, cumin, turmeric, garlic, and ginger can effectively prevent protein denaturation during frozen storage. Additionally, they suggested that incorporating cross-linking into the protein structure of egg gel can effectively reduce drip loss during frozen storage. Furthermore, Rachman et al. (2020) have suggested that OEx4 demonstrates a protective effect against drip loss caused by the thawing of sausages that have been stored in a frozen state. This indicates that OEx4 may offer a potential solution to minimize the negative effects of frozen storage on the quality of chicken meat products.
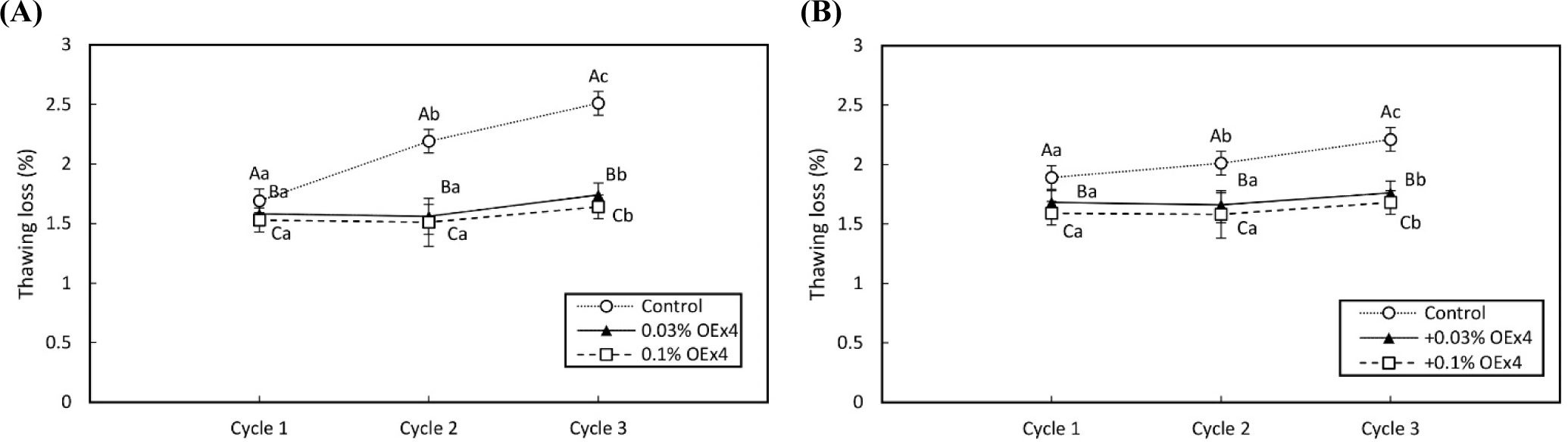
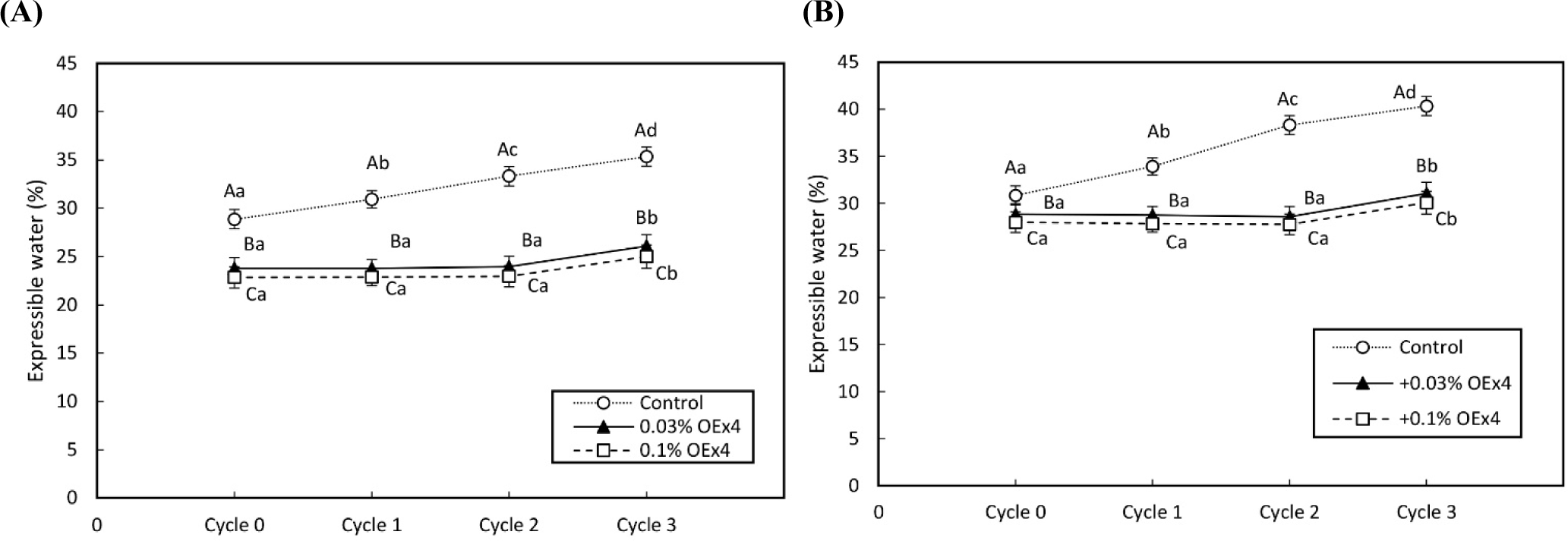
Fig. 3 shows changes in the expressible water by repeating the freeze-thaw cycle. In both WE and EW gels, the expressible water of the control gel sample increased (p<0.05) with each cycle. This implies that repeating freeze-thaw induces a decrease in bound water volume concomitant with the increase in the volume of free water (Lv and Xie, 2021). On the other hand, the expressible water of the two gel samples containing 0.03% and 0.1% OEx4 was little changed (p<0.05) by freeze-thaw. This suggests that bound water in OEx4-gel strongly interacted with the frame of gel, even if the freeze-thaw of gel was repeated. As a result, bound water was maintained in its gel matrix. Furthermore, the effect was seen in WE gel which contains lipids as well as EW gel without lipids, suggesting that the high water-holding effect in OEx4 gel is applicable in complicated food gel systems that contain multiple types of ingredients. In a study by Moreno et al. (2010), it was discovered that combining alginate and microbial transglutaminase (MTGase) can prevent a decrease in WHC during freezing storage. Additionally, different saccharides interact with egg protein, reducing water loss during freeze-thaw cycles and delaying the decline in WHC of heat-induced egg gel during freezing (Chen et al., 2022; Zhang et al., 2023). OEx4 also causes crosslinks between proteins, contributing to high freeze-reliance. Furthermore, olive leaf extract, which has a high polyphenol content, possesses potent antioxidant properties, such as scavenging hydroxyl (OH⋅), superoxide (O2⋅), and DPPH radicals, as well as chelating ferrous ions (Fe2+). By incorporating polyphenols into eggs before frozen storage, it is possible to prevent the formation of protein carbonyls and dityrosine, reduce the loss of surface hydrophobicity in egg proteins, and enhance the total sulfhydryl content, resulting in less protein degradation and a more intact egg protein structure.
Despite the presence of OEx4 in egg gels, repeated freeze-thaw cycles of egg gel induced syneresis to a small degree. Herein, we evaluated how much the syneresis affects pH (Tables 3 and 4). In both of WE and EW gels, the pH value of the control gel increased in second cycle of freeze-thaw (p<0.05), while that of OEx4 gels did not change much (p>0.05). When the pH exceeds 7, the protein charge increases, resulting in the formation of a three-dimensional gel by ovalbumin (Ferreira Machado et al., 2007). Gels induced by strong bases primarily rely on ionic and disulfide bonds. Raikos et al. (2007) observed a direct correlation between gel strength and pH for thermally induced whole eggs and EY protein, and a relatively small linear relationship between gel hardness and pH for EW protein. Additionally, these findings suggest that using OEx4 in protein-based gel food helps maintain the food’s pH stability during frozen storage. However, due to the instability of OEx4 in an alkaline environment, quinonoids were formed when OEx4 was added to EW. For instance, Ai et al. (2019) reported that quinonoids have a strong binding capacity with proteins and promote nonspecific cross-linking between proteins and other molecular interactions, including ionic bonds, hydrophobic interactions, and disulfide bonds.
The initial cycle revealed that the microbial contamination of egg gel did not surpass 3 Log CFU/g in any of the OEx4 and control samples (p>0.05). This indicates that such a level of contamination is common, as other studies have also shown (Cwiková and Nedomová, 2014; Necidová et al., 2019). The olive leaf extract had diverse effects on the TPCs of egg gel. Although the TPCs were initially similar, they varied significantly over the following weeks. In the third cycle of freeze-thawing, TPCs in egg gel with contained olive leaf extract stayed below 3 Log CFU/g, while the control without the olive leaf extract showed bacterial growth up to 3–5 Log CFU/g (Tables 5 and 6). Interestingly, the egg gel control samples without olive leaf extract had the highest TPCs by the end of the observation period (cycle 3). Coliform bacteria are commonly utilized as bacterial indicators to assess the hygienic quality of eggs and egg products (Chousalkar et al., 2021). The absence of coliforms in this case is likely due to the high quality of the eggs and the hygienic processing methods employed in both the egg gel with OEx4 and the control group. The olive leaf extract has been shown to enhance the quality and shelf-life of meat products (Alirezalu et al., 2016; Hayes et al., 2010). Also, Takó et al. (2020) suggested that phenolic compounds derived from plants, including phenolic acids, flavonoids, stilbenes, and tannins, have been shown to inhibit the growth and activity of a variety of microorganisms in frozen food. This includes food-related pathogens and clinically significant bacteria, fungi, and protozoa. These findings highlight the effectiveness of the olive leaf extract in inhibiting bacterial growth and reducing contamination in egg gel.
Figs. 4 and 5 demonstrate the changes in gel strength of OEx4 egg gels during freeze-thaw cycles for WE and EW gels. The breaking stress of the control gel sample without OEx4 decreased significantly (p<0.05) after repeated freezing and thawing. In contrast, the breaking stress of OEx4-containing gels remained stable (p>0.05) in the first and two cycles, with only a slight decrease (p<0.05) of 6% to 9% in the third cycle. The viscosity characteristics of the OEx4-egg gels following freeze-thaw were evaluated using elongation and stability analysis. Fig. 6 illustrates changes in the elasticity of WE and EW gels. The elasticity of control samples without OEx4 significantly decreased (p<0.05) with repeated freeze-thaw, with a more significant change in WE gel than EW gel. Conversely, the elasticity of the gel samples containing 0.03% and 0.1% OEx4 remained unchanged (p>0.05) until the third freeze-thaw cycle, where a minimal reduction was observed compared to the control gel. Changes in viscosity parameters are presented in Fig. 7, showing a significant decrease (p<0.05) in the viscosity of control gels with repeated freeze-thaw. In contrast, the viscosity of OEx4-gels changed minimally throughout the freeze-thaw cycle. The addition of polyphenols is an efficient method to prevent damage to the physical, chemical, and functional properties of proteins during the freeze thaw cycle (Cao et al., 2020). Additionally, hydrogen bond interactions between the protein amino groups and polyphenol hydroxyl groups favor aggregate formation, improving protein stability. Also, the abundant phenolic hydroxyl groups of polyphenols confer high affinity for metal ions, which prevents the various biochemical reactions triggered by these cofactors (Zhu et al., 2021).
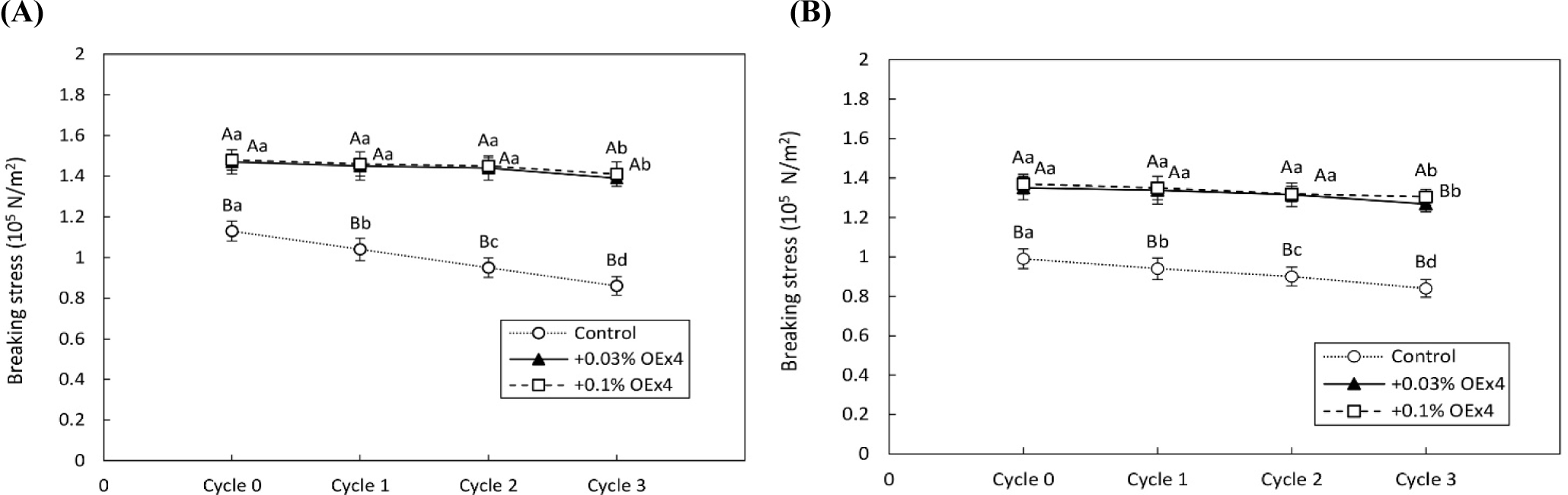
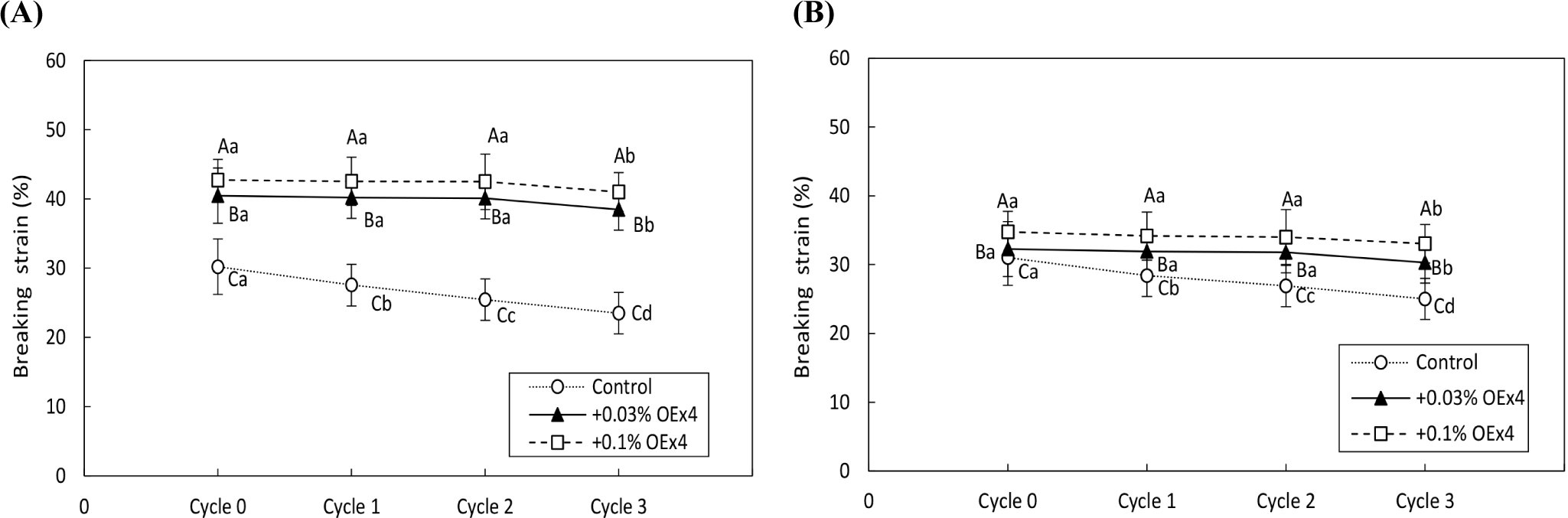
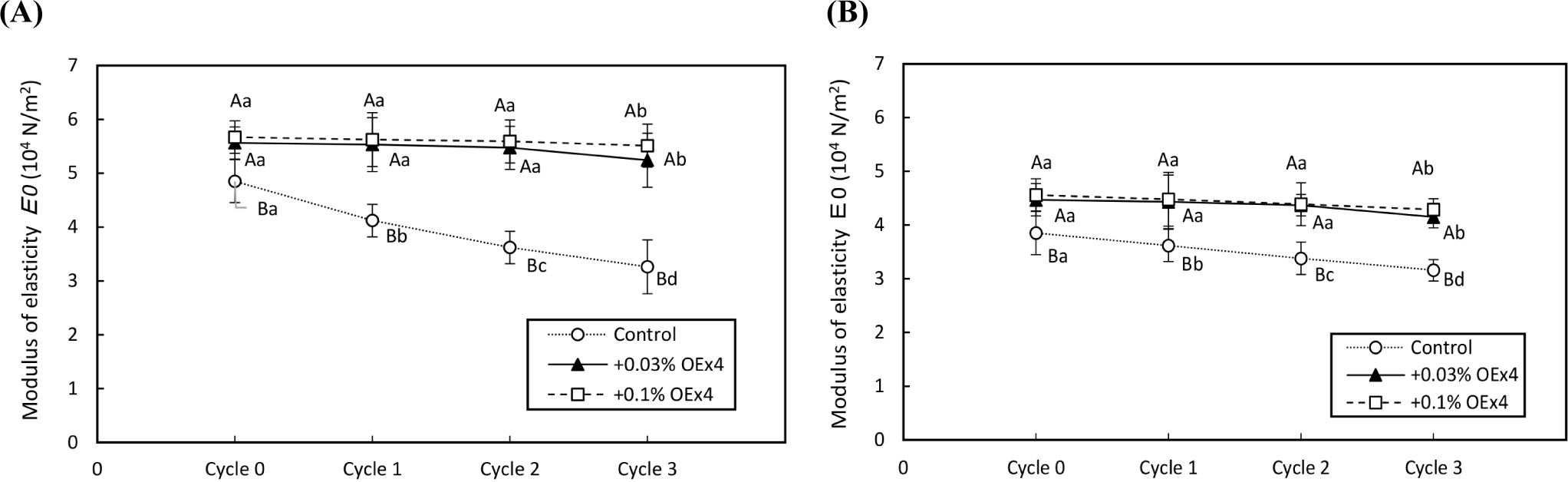
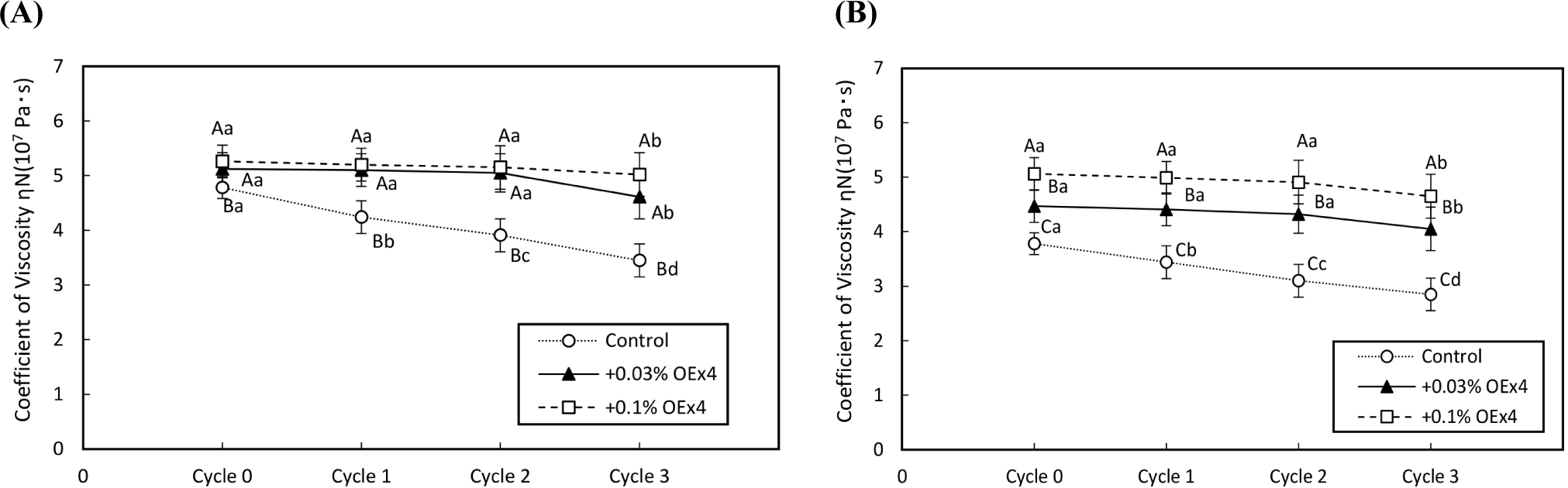
Fig. 8 illustrates the microstructure of unfrozen and frozen WE gels, revealing a rough texture with large voids in the frozen gels, especially the control gel. However, no such voids were observed in the 0.1% OEx4-gel. Fig. 9 depicts the microstructure of freeze-thawed EW gels, showing a rough surface with large voids in the control gel, while the 0.03% OEx4-EW gel was smooth with no large voids but slightly rougher. No such voids were observed in 0.1% OEx4 gels subjected to freeze-thaw cycles. This microstructure of OEx4-egg gels is similar to the microstructure of sodium chloride- EY gels during frozen storage, when sodium chloride was added to the EY gels, more proteins were released from granules and participated in gel formation, this yielding a more compact microstructure (Liu et al., 2018). This result is consistent with the conclusion obtained from Figs. 6 and 7 that freezing and thawing treatment contributed to a stronger network of elasiticity and viscosity OEx4-egg gels than that of control gels. Likewise, the strong cross-linking of OEx4-egg gels were also shown greater strength than that of the control gels during freeze thaw cycle (Figs. 4 and 5). The minimal changes in the morphology of OEx4-egg gels suggest their ability to withstand freeze-thaw cycles, as evidenced by their strength and creep analysis results. The strong freeze resistance of OEx4 gels may be attributed to the stabilization of water behavior in the EY.


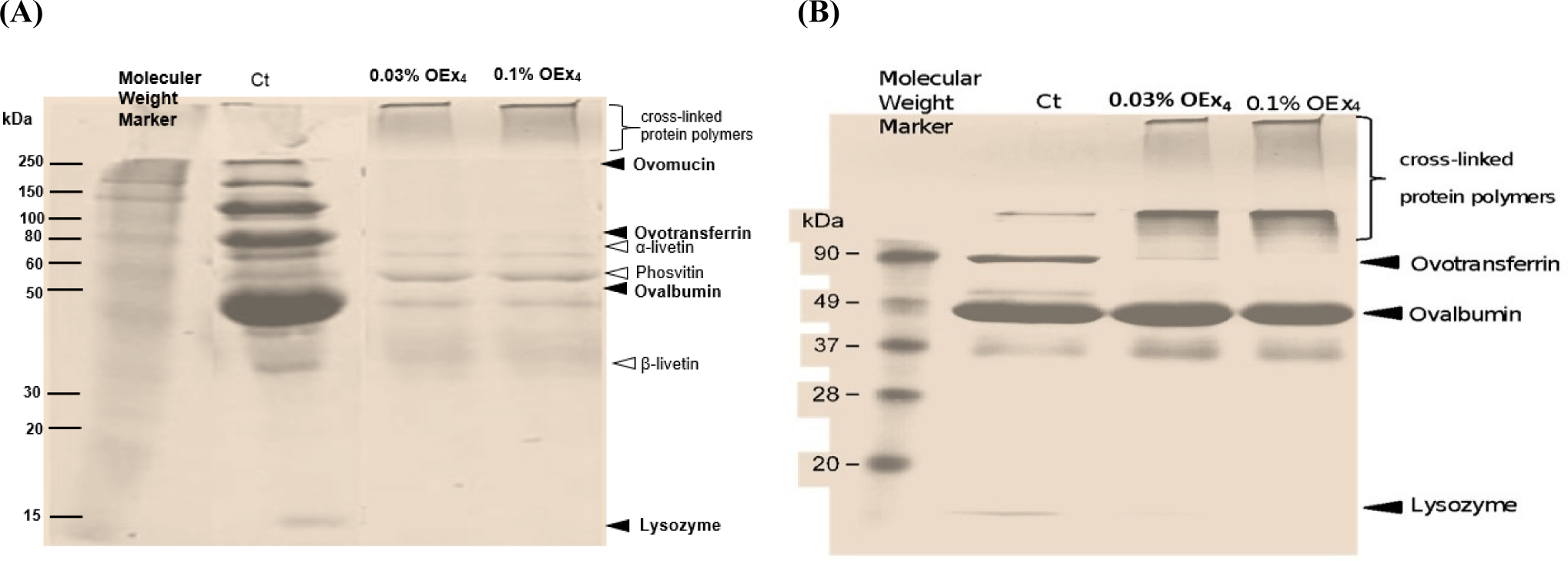
The SDS-PAGE pattern of the protein extraction from the heat-treated OEx4-WE solution is shown in Fig. 10A. The remaining bands were thinner compared to the control WE protein. Specifically, α-livetin and ovotransferrin disappeared, while ovalbumin, lysozyme, phosvitin, and β-livetin became thinner. A new high molecular weight band emerged, indicating the formation of a protein complex due to the inclusion of OEx4 (Rachman et al., 2021). Intense bands at over 250 kDa were observed at the top of the separating gel, comprising high-molecular-weight crosslinked proteins (Xue et al., 2022). This indicates non-disulfide-type intermolecular crosslinking among these proteins, resulting in polymer formation. In the SDS-PAGE pattern of the heat-treated OEx4-EW protein solution (Fig. 10B), the prominent 75 kDa ovotransferrin bands were no longer visible. Instead, intense bands over 90 kDa were observed at the top of the separating gel, containing high-molecular-weight cross-linked proteins, including ovalbumin and lysozyme. Ovalbumin bands in OEx4-EW gels indicate the formation of a network structure through covalent crosslinking. Wang et al. (2024) discovered that tea polyphenols can shield EW proteins from damage in alkaline conditions by acting as crosslinking agents, with ovalbumin playing a crucial role. Additionally, tea polyphenols can modify the environment around the crosslinked proteins, making EW proteins more water-attracting and preventing protein damage, ultimately enhancing the formation of network proteins in EW gels.
To investigate the functional groups of egg proteins involved in protein polymerization, Akazawa et al. (2021) analyzed the proteins’ sulfhydryl and primary amino groups. The formation of ortho-quinone through the oxidation of phenolic compounds can lead to the binding of nucleophiles, such as amino or sulfhydryl groups of proteins. This can result in the formation of C–N or C–S bonds, as discussed by Cao and Xiong (2015), Jongberg et al. (2011), and Liu et al. (2017). Akazawa et al. (2021) found that combining olive leaf with EW protein leads to changes in certain chemical groups, forming larger protein molecules. Adding 0.2 grams of olive leaf per gram of EW protein causes a 24% decrease in primary amino groups and a 77% decrease in sulfhydryl groups. The researchers also observed a significant 97% reduction in a compound called oleuropein aglycone or decarboxymethyl oleanolic acid linked to hydroxytyrosol (3,4-DHPEA-EDA) at 49.2 min when the compounds were exposed to heat. This compound contributes to the olive leaf extract ability to bind proteins together.
Conclusion
The application of OEx4 was highly effective in preventing egg gels’ physical and microbiological deterioration when subjected to freeze-thaw cycles. Thus, OEx4 helps improve the storage stability of frozen egg products. This effect probably is due to the insertion of cross-linkage into the egg protein gel network. The cross-linkage raised the WHC of the gel and the robust gel state formation, as a result, the gel acquired high frozen tolerance.













